 If electronegativity values aren't given, you should assume that a covalent bond is polar unless it is between two atoms of the same element. So you can fit a maximum of two electrons in one orbital. So in general, we can predict that any metal-nonmetal combination will be ionic and any nonmetal-nonmetal combination will be covalent. Lets move on to electrons in the orbital. Metals have low electronegativity compared to nonmetals. If you are not given electronegativity values, you can still predict the bond type using the periodic table. Because each orbital is different, they are assigned specific quantum numbers: 1s, 2s, 2p 3s, 3p,4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p. Each has its own specific energy level and properties. If the difference is between 0.4 and 2.0 units, the bond is classified as polar covalent, and if the difference is more than 2.0 units, the bond is substantially ionic. Quantum Numbers describing Electronic Orbitals There are multiple orbitals within an atom. If the electronegativities differ by less than 0.4 units, the bond can be classified as nonpolar covalent. One method to classify bonds based on this difference can be described as follows. On the other hand, the fourth quantum number does not tell us where the electron is, but how. Although n can be any positive integer, only certain values of l and ml are allowed for a given value of n. Three of them give us information about where an electron of a certain atom is, that is, they give us information about the orbital. The quantum numbers provide information about the spatial distribution of an electron.  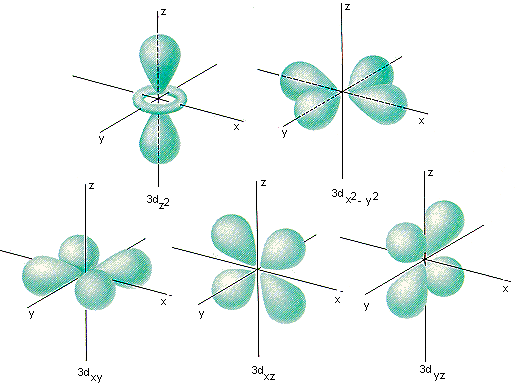
Electronegativity difference can be used to predict bond type. 3 Exercises and examples What are the quantum numbers There are 4 quantum numbers. An ionic bond results when the sharing is so unequal that fully charged ions form. A polar covalent bond is one in which the electrons are unequally shared between the atoms. Each electron has its unique set of quantum numbers, which means that two electrons can share one, two, or even three quantum numbers, but never all four. 
When two bonded atoms attract electrons with equal strength, the result is a nonpolar covalent bond. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
