 
The sample of the upper part of the solution was withdrawn with a suitable warmed pipette to another weighed vial. Then, the excess solid could be observed in the lower part of the equilibrium cell. After 48 h, the stirring was stopped and the solution was kept still for 48 h. Then, the equilibrium cell was heated to the required temperature with continuous stirring. For each measurement, an excess amount of caffeine was added to a known mass of solvent. 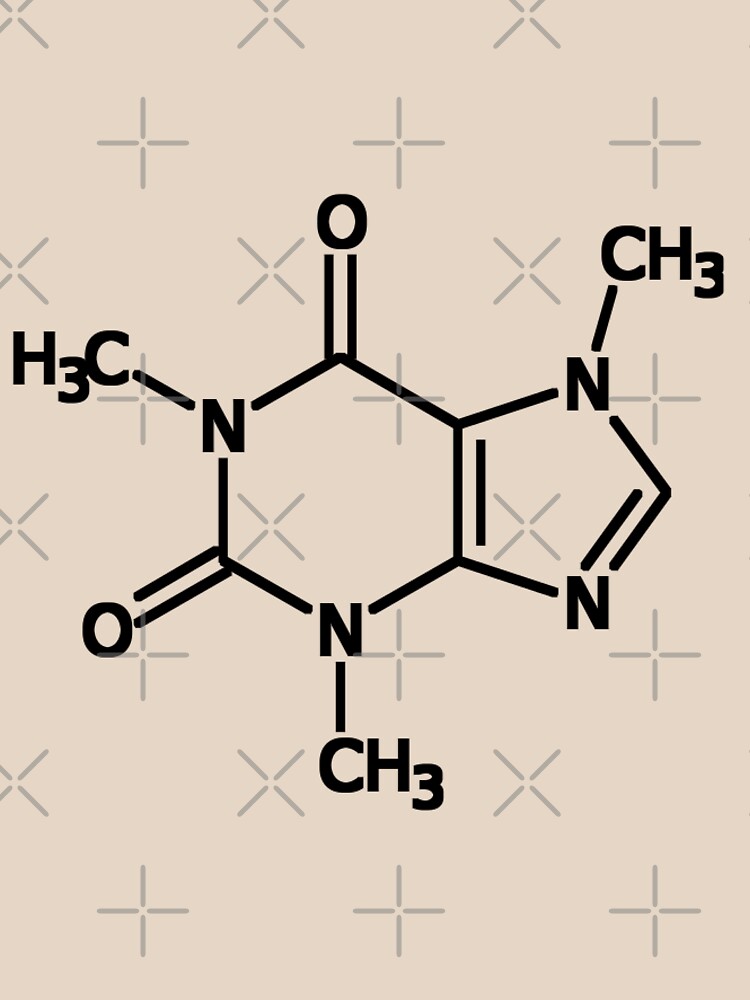
To ensure equilibrium, undissolved solid and solution were allowed to settle for 48 h before sampling. For each measurement, an excess of known mass of caffeine was added to a known mass of solvent. The solubilities of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane and acetone were measured by the gravimetric method which reported by early investigator (Zhu, 2001). All the chemicals were used without further purification. The organic solvents, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane and acetone were of analytical purity grade and were purchased from Merck (Darmstadt, Germany) and re-distilled deionized water was used. The experimental solubility of caffeine in water, ethanol and ethyl acetate was compared with literature data which were reported earlier (Bustamante et al., 2002).Ĭaffeine with mass fraction purity of > 98.5 % (Merck, Darmstadt, Germany) was used as obtained. In this study, the solubilities of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane and acetone have been measured from (298 to 323) K at atmospheric pressure by a gravimetrical method and the solubility data was correlated against temperature. So it is necessary to know the solubility data of caffeine in water and organic solvents (such as ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane and acetone etc). Recent results (El-Din and Abu-Raiia, 1995 Treybal, 1968) showed that the extraction processes depend upon the plants and solvents selected. Caffeine extraction is an important industrial process and can be performed using a number of different solvents. The method is based on extraction from the filtrates of water-plant systems by means of solvents. Generally, caffeine is extracted from plants such as cacao, tea and coffee. Overall, caffeine is found in the beans, leaves, and fruit of over 60 plants, where it acts as a natural pesticide that paralyzes and kills certain insects feeding upon them. It is found in the leaves and beans of the coffee plant, in tea, yerba mate, and guarana berries, and in small quantities in cocoa, the kola nut and the Yaupon Holly. Caffeine is the world's most widely consumed psychoactive substance. Beverages containing caffeine, such as coffee, tea, soft drinks and energy drinks enjoy great popularity. Early investigators (Gürü and İçen, 2004) indicate that caffeine is a central nervous system (CNS) stimulant, having the effect of temporarily warding off drowsiness, restoring alertness and muscle relaxant properties. As with all methylxanthines, caffeine has low solubility and is therefore often combined with a wide variety of compounds to form complexes, such as the double salt sodium benzoate, for purposes of enhanced solubility in consumer goods like soft drinks. In its pure form, caffeine occurs as odorless, white, fleecy masses, glistening needles or powder. It belongs to the group of chemicals known as methylxanthines, which also includes the closely related chemicals theophylline and theobromine. Correlation.Ĭaffeine is an alkaloid, or nitrogen-containing substance, with the chemical formula C 8H 10N 4O 2. The solubility of caffeine in chloroform and dichloromethane was high compared with other solvents Solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane, and acetone between 298 and 323 KĬhemical Industries Department, Iranian Research Organization for Science and Technology (IROST), Tehran, - The solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane and acetone were measured by a gravimetrical method from (298 to 323) K and the solubility data was correlated against temperature. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
