 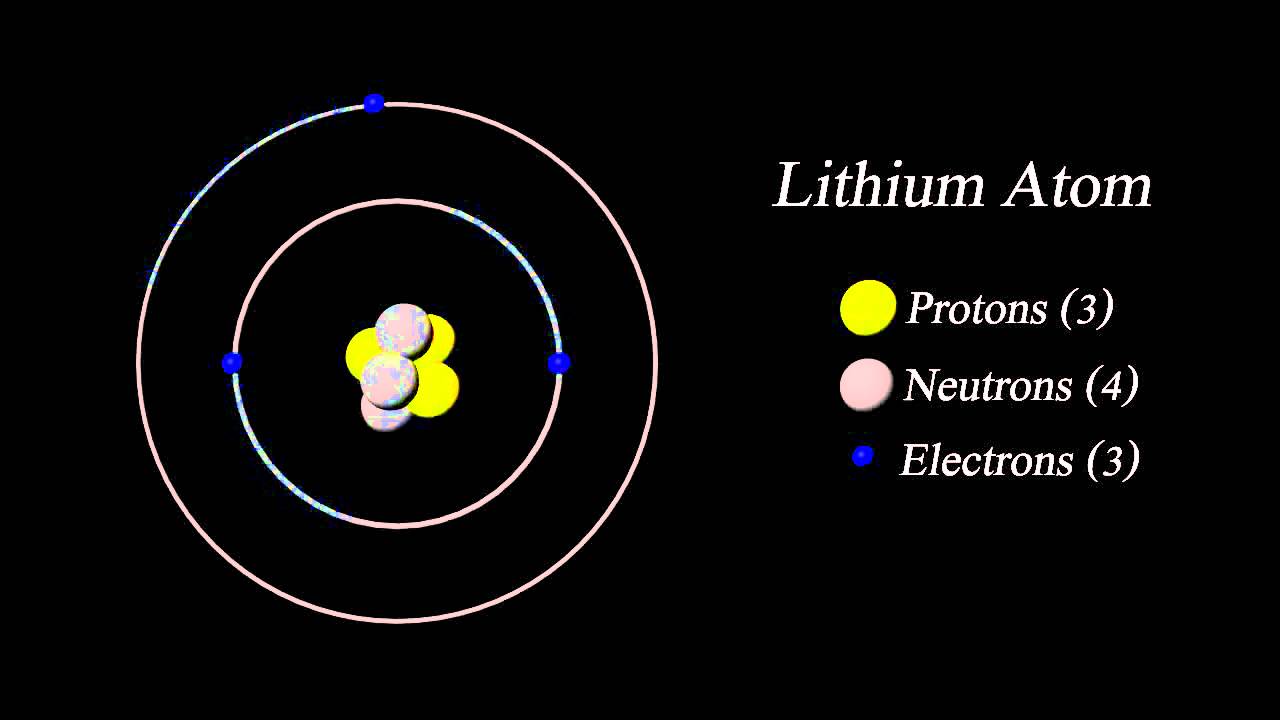
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. Is the atomic mass of lithium 6 or 7 The average atomic mass of lithium is 6. Under standard conditions, it is the least. The atomic number of Lithium is 3, its mass number is 7. It is a soft, silvery-white alkali metal. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. Lithium is a chemical element with the symbol Li and atomic number 3. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. It has the atomic symbol Li, atomic number 3, and atomic weight 6.938 6.997. Lithium – Properties Summary Element Lithium Atomic Number 3 Symbol Li Element Category Alkali Metal Phase at STP Solid Atomic Mass 6.941 Density at STP 0.535 Electron Configuration 2s1 Possible Oxidation States +1 Electron Affinity 59.6 Electronegativity 0.98 1st Ionization Energy 5.3917 Year of Discovery 1817 Discoverer Arfvedson, Johan August Thermal properties Melting Point 180.5 Boiling Point 1342 Thermal Conductivity 85 Specific Heat 3.6 Heat of Fusion 3 Heat of Vaporization 145.For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. Lithium-7 atom is the stable isotope of lithium with relative atomic mass 7.016004, 92.5 atom percent natural abundance and nuclear spin 3/2. The Standard English unit is pounds mass per cubic foot ( lbm/ft 3). The standard SI unit is kilograms per cubic meter ( kg/m 3). In other words, the density (ρ) of a substance is the total mass (m) of that substance divided by the total volume (V) occupied by that substance. It is an intensive property, which is mathematically defined as mass divided by volume: Typical densities of various substances at atmospheric pressure.ĭensity is defined as the mass per unit volume. of the Following Atoms Showing the Nucleus Containing Protons, Neutrons and the Orbits with the Respective Electron: Lithium At. How does the atomic mass determine the density of materials? Density of Lithium The atomic mass number determines especially the atomic mass of atoms. The mass number is different for each different isotope of a chemical element. For 63Cu, the atomic mass is less than 63, so this must be the dominant factor. A nucleus with greater binding energy has lower total energy, and therefore a lower mass according to Einstein’s mass-energy equivalence relation E = mc 2. 
The nuclear binding energy varies between nuclei. 
This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
